The amount of electrons in a molecule affects how tight the nuclear charge can control the overall charge distribution. 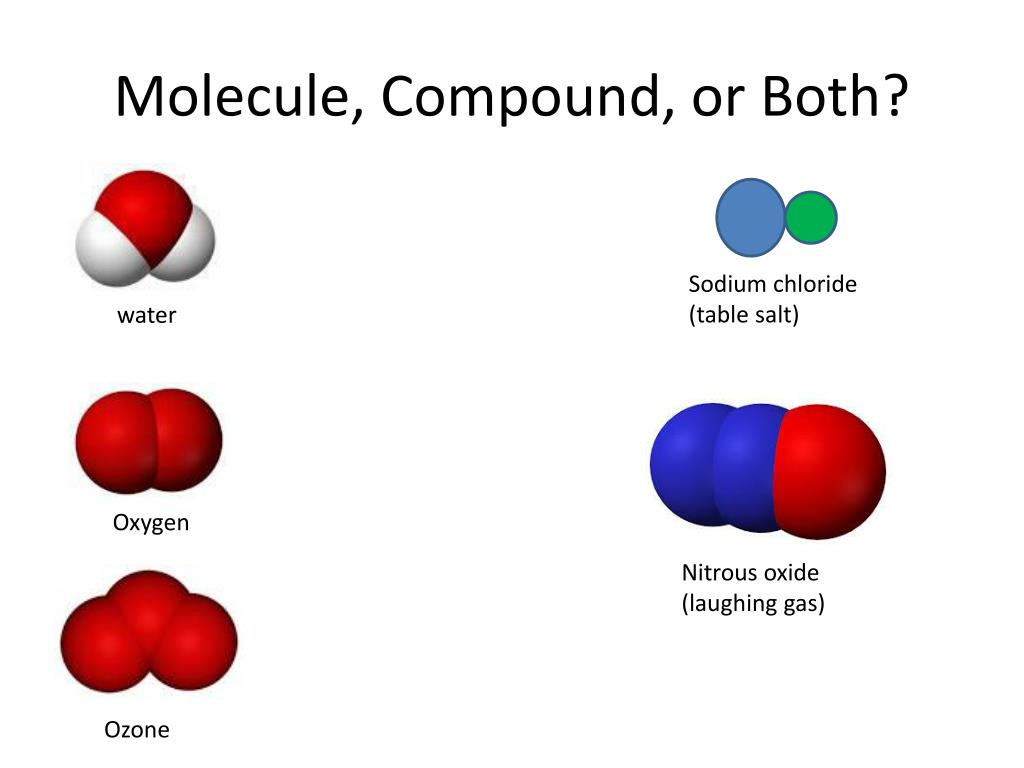
In general, polarizability correlates with the interaction between electrons and the nucleus. Pure substance chemical properties What is not a pure substance? C 12 H 22 O 11 , and a pure substance that is a compound but not considered a molecule is sodium chloride, N a C l NaCl N a Cl (table salt). If we turn off the electric field, the dipole moment goes away (not shown). This is a partial list of physical properties that can be observed, measured, and predicted for pure substances under normal conditions (room temperature and one earth atmosphere of pressure): Sodium chloride from the Dead Sea is physically the same as sodium chloride mined in Kansas. Pure substances have a constant composition. Learn the definition of diatomic elements, where to find diatomic elements on the periodic table, how diatomic elements are formed, and how to remember them. No matter where a given pure substance is found, it will have identical physical properties to every other pure substance of the same chemical makeup. A molecule is a group of two or more atoms chemically bonded to each other and is neutral. These predictable properties make pure substances the foundation of chemistry because known interactions can be used to form new materials and new types of matter. The definition is used to devise a tailored periodic. According to the periodic table, hydrogen has. It is shown that the current periodic system of chemical elements is an instance of the general structure. He had created the periodic table, making it possible to predict how any. Such bonds are directional, meaning that the atoms adopt specific positions relative to one another so as to maximize the bond strengths. (more) Molecules are held together by shared electron pairs, or covalent bonds. Whether element, molecule, or compound, pure substances share similar physical and chemical properties. The periodic table of elements can give us a wealth of information about atoms, and we will use it to help us understand hydroxide's chemical formula. A ball-and-stick model of molecular structure, showing atoms bonded together. Pure substance definition Properties of pure substances
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
